Quality Control is What MakesYour Beverage a Quality Product
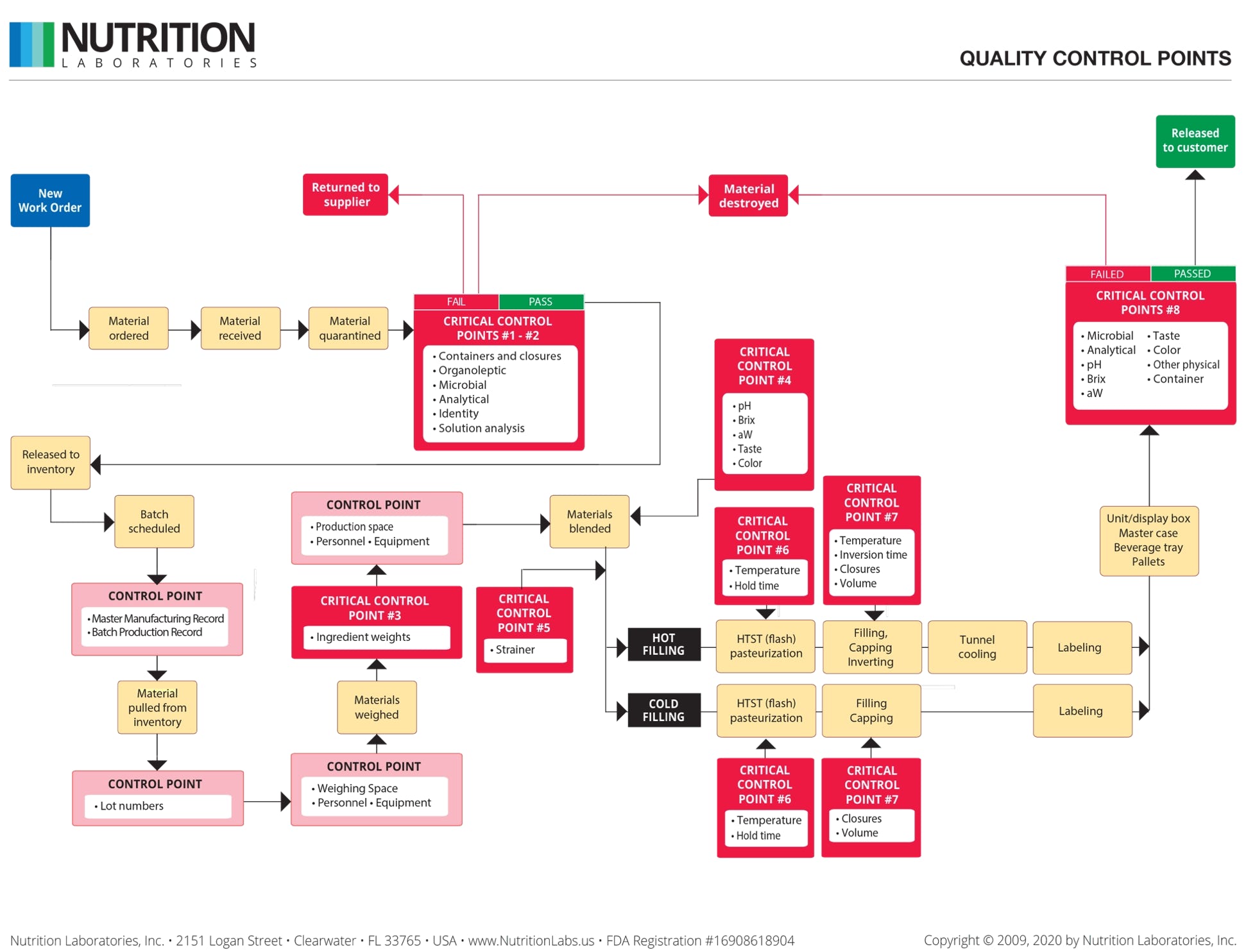

Qualty Control at Each Step.
QC inspectors monitor every step of production to ensure your beverage meets all specifications and remains shelf-stable for at least 24 months.
Allergen Control Program.
We keep allergen-containing ingredients—such as whey protein (milk) and coconut water (tree nuts)—segregated to prevent cross-contamination. After producing a beverage with these ingredients and completing equipment sanitization, a QC inspector swabs tanks, hoses, and fillers to verify that no allergen proteins remain.
Licenses and Certifications
FDA and State of Florida Registrations.
Nutrition Laboratories is licensed by the FDA to produce beverages as food products or dietary supplements. We are also registered by Florida Department of Agriculture. Both the FDA and State of Florida inspect the company for compliance with Good Manufacturing Practices.
NSF Certification.
In addition to the FDA and State of Florida, NSF conducts annual audits to verify that we comply with Good Manufacturing Practices.
Organic Manufacturing.
Nutrition Laboratories is a certified organic manufacturer with Americert International. If your product qualifies, we will get it approved as a certified organic beverage.
Kosher Manufacturing.
If you want your product to be certified as Kosher, Atlanta Kashruth Commission will certify it for you. We have been approved by AKC as a Kosher manufacturer.
Registered Florida Hemp Producer.
Nutrition Laboratories is licensed by the State of Florida to produce beverages with hemp CBD.
Non-GMO Manufacturing.
We have been approved by the Non-GMO project to bottle non-GMO beverages.
Conventional Beverages & Dietary Supplements

3rd Party cGMP Certification
© 2025 by Nutrition Laboratories, Inc. All Rights Reserved | Privacy Policy